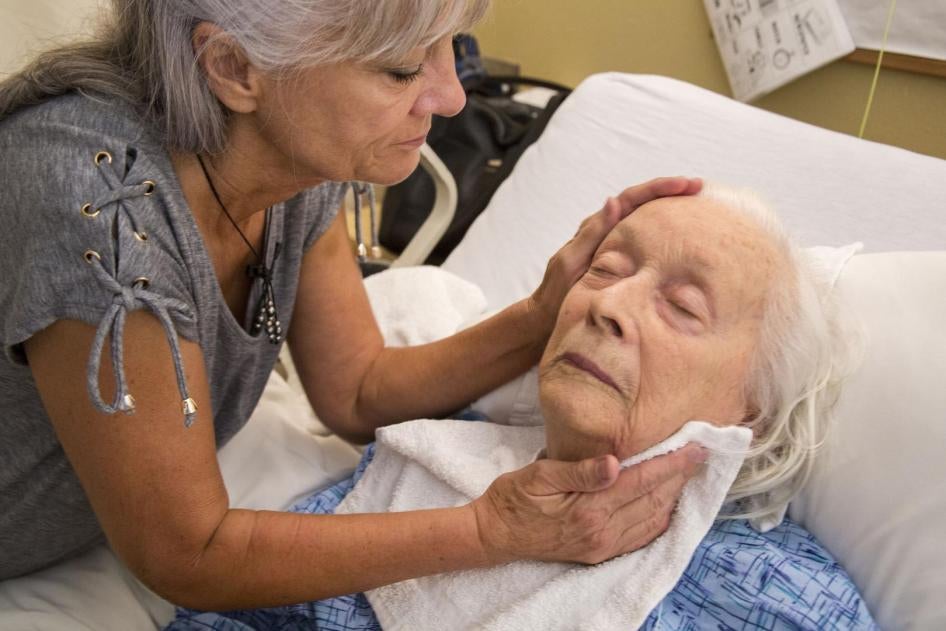
A year and a half ago in a Texas nursing home, I met an 84-year-old resident with dementia named Felipa Natividad. Her sister, Aurora Suarez, told me that the staff dosed Natividad with Haldol, an antipsychotic drug, to ease the burden of bathing her. “They give my sister medication to sedate her on the days of her shower: Monday, Wednesday, Friday,” Suarez said. “They give her so much she sleeps through the lunch hour and supper.” A review of Natividad’s medical chart confirmed the schedule.
Suarez said she had given her consent to use the drugs because she feared that the staff would not bathe her sister enough if she refused. But when Suarez saw the effect they had, she had second thoughts. She expressed them to the nursing home, but Natividad was taken off the antipsychotics only after she was placed in hospice care. She died a few months after my interview. Her family, seeing her in a reduced state and unable to communicate, wondered whether the drugs had compounded the losses associated with dementia; Suarez thought they contributed to her sister’s decline. “She gets no nourishment,” she told me not long before Natividad died.
The use of antipsychotic drugs as chemical restraints — for staff convenience or to “discipline” a resident — has a long history in nursing homes. In 1975, the Senate released a report, “Drugs in Nursing Homes: Misuse, High Costs, and Kickbacks,” documenting some of the same trends we still see, more than 40 years later. In the past decade, many manufacturers of antipsychotic drugs have faced civil and criminal penalties for misbranding the medications to promote them as appropriate for treating older people with dementia. For more than a decade, the Food and Drug Administration has required manufacturers to place the strongest caution, known as a “black box warning,” on the packaging to advise against the medicines’ use in these patients; such drugs almost double the risk of death for them and have never been approved as safe oreffective for treating symptoms of dementia. Despite the warning, nursing homes still often administer antipsychotic drugs in this manner, sometimes without seeking informed consent first, in violation of federalregulations and human rights norms.
Last year, I visited more than 100 nursing homes across six states as part of Human Rights Watch research on the abuse of antipsychotic drugs in such facilities. Based on government data, we estimate that in an average week, more than 179,000 older people in nursing homes are given antipsychotic drugs without an appropriate diagnosis. The powerful medications were developed to treat schizophrenia, but staff most commonlyadminister them to older people with dementia. Too often, nursing homes use the antipsychotics for their sedative effects rather than to treat a medical problem. It’s true that the prevalence of antipsychotic drugs has declined in recent years, from 1 in 4 nursing home residents without an approved diagnosis in 2012 to 1 in 6 today, but that falsely suggests that a longtime wrong is being righted. Government enforcement of regulations prohibiting the use of the drugs as a chemical restraint or without informed consent remains weak. What’s more, two Trump administration decisions threaten the progress made in curbing the abuse.
These powerful drugs are misused for a variety of reasons, including a misperception by nursing home staff that the medications can help people with dementia; a lack of awareness of their dangers, despite the black box warnings; lack of training in dementia care; and, perhaps most significant, to compensate for understaffing. Nursing homes have been exaggerating levels of nursing and caretaking staff for years, according to an analysis of federal data by Kaiser Health News.
To understand the human toll of the misuse of antipsychotics, in my visits to the nursing homes I interviewed more than 300 people — residents, their families, staff, ombudsmen and doctors, as well as researchers and regulators. In most cases, I did not use their names in my report because they cited a fear of retaliation for speaking openly to me. Human Rights Watch did not identify the individual facilities in its report because the abuse is so pervasive in the industry, and not identifying nursing homes persuaded some staffers to speak with me. Further, the goal was not to expose particular private actors so much as to pressure government officials to enforce minimum health and safety standards for all providers.
I found that too often, antipsychotic drugs are administered in harmful, avoidable ways and without the appropriate consent. They are used to control people. Nursing home residents and their family members repeatedly told me they were given these medicines without their knowledge, without awareness of the risks or over their objections. Staff members frankly admitted giving residents these medicines for their own convenience, with some saying they were not aware of an informed-consent policy. Nursing facility staff, pharmacists and medical directors described how doctors commonly prescribed the medications at the request of nursing staff, without even seeing the patients.
[How corporate funding can distort NIH research]
Nursing homes turn to antipsychotic drugs — among other classes of psychotropic medications — because dementia is associated with agitation, irritability, aggression, delusions, wandering, disinhibition and anxiety. While such symptoms are frightening for the people experiencing them and challenging for their caregivers, institutional or otherwise, antipsychotic drugs have not been found to be effective at managing them. In a small number of particularly complex cases, antipsychotic drugs may be appropriate as a last resort. But that is a far cry from how they are used. The American Psychiatric Association concludes that the drugs offer “at best small” potential benefits (such as minimizing the risk of self-harm in people with extreme agitation), while “on the whole, there is consistent evidence that antipsychotics are associated with clinically significant adverse effects, including mortality.”
On paper, federal regulations say that residents have the right to be fully informed of their treatment and to refuse treatment, which should amount to a right to informed consent. But nursing facilities widely ignore the rules, partly because they are rarely held accountable. Reviewing government data from 2014 through mid-2017, Human Rights Watch found that in 97 percent of citations for violations related to antipsychotic drugs, the incidents were determined to have caused “no actual harm” to residents. As a result, in almost no cases did the government impose financial penalties, which correspond to the severity of harm caused by the noncompliance. The prospect of enforcement actions, and the rare sanctions issued, unsurprisingly had little deterrent effect, our analysis found. Nursing homes cited for antipsychotic-drug-related issues did not reduce their rates of drug use any more than facilities not cited.
The way nursing homes obtain or define “informed consent” can also be a factor in the drugs’ misuse. “The use of specific medicines, particularly for somebody with dementia, who lacks the capacity to consent themselves, should require informed consent from their legal representative,” says Jonathan Evans, a former president of the American Medical Directors Association. “But in practice that seldom happens. Not just for that medicine but for any medicine.”
The former administrator of a nursing home in Kansas, who asked to remain anonymous, said: “The facility usually gets informed consent like this: They call you up. They say: ‘X, Y and Z is happening with your mom. This is going to help her.’ Black box warning? It’s best just not to read that. The risks? They gloss over them. They say, ‘That only happens once in a while, and we’ll look for problems.’ We sell it. And, by the way, we already started them on it.”
Despite the limited threat of penalties, many nursing homes have reduced inappropriate use of the drugs in recent years anyway, in response to increased public pressure. By 2012, congressional outrage over the widespread misuse of medications, costing hundreds of millions of Medicare dollars annually, had motivated the Centers for Medicare and Medicaid Services (CMS) to create the National Partnership to Improve Dementia Care in Nursing Homes . While the partnership is only a voluntary initiative offering educational support to train providers, the increased attention has been associated with the reduction in misuse. But it is hard to celebrate the decline when the government elects not to rigorously use the tools it has to protect Americans in nursing homes from irreversible harm.
What’s more, the national reduction may be misleading — and it may not last. First, there’s been a notable uptick in the diagnosis of schizophrenia (a disease that typically develops before age 30) in predominantly elderly nursing home populations. This increase corresponds to the rising pressure on facilities to reduce off-label antipsychotic drug use. There’s no proven link, but the trend does suggest that some homes seek false diagnoses to avoid red flags with the use of these medications. A second concern, recognized by CMS, is that nursing homes are simply replacing this closely watched class of psychotropic medication with other types of sedating drugs with similar health risks.
[Aging is not death. Stop conflating the two.]
Meanwhile, in response to an industry request, the Trump administration in July 2017 changed its guidanceon financial penalties, limiting the instances when inspectors can assess the heaviest fines. The guidance also favors one-time sanctions for harmful noncompliance with the law, rather than a per-day sanction that corresponds to the number of days the harm persists. As a result, in many cases facilities face less-significant consequences for harming residents than they used to. And last November , CMS imposed an 18-month moratorium on Obama-era revisions to some regulations — not updated since 1991 — intended in part to protect residents whose psychotropic medications are prescribed on an “as needed” basis. While it is unclear if those new protections will come into force, it is abundantly clear that this administration’s deregulatory scheme, which it calls “Patients Over Paperwork,” reduces oversight and enforcement in an already dangerously underpoliced industry.
Karla Benkula, daughter of a 75-year-old woman in Kansas, said that when the nursing facility began giving her mother an antipsychotic drug, her mother “would just sit there like this. No personality. Just a zombie.” Laurel Cline, the daughter of an 88-year-old woman in a California nursing home, said she thought the facility used antipsychotic medication to silence people whose symptoms disturbed the staff. Cline said it was obvious that her mother had physiological conditions requiring medical attention. “She would be sitting there, slumped over, mucus everywhere. I would go over and say, ‘She’s sick.’ ” But Cline’s mother wasn’t able to advocate for herself, and Cline had to intervene to demand appropriate medical care for a urinary tract infection, pneumonia and a pulmonary embolism, she said.
“Dementia’s already so hard,” said Ashley Plummer, a licensed practical nurse who works in a Kansas nursing home. “But on top of that, throw [on] a few Seroquels [a common antipsychotic drug], and then you’re just drooling. I mean, it’s taking away even your right to be upset about your disease process. It’s taking away your right to mourn what you’re going through.”
In my interviews, it was disturbing how frequently staff justified administering antipsychotic drugs for “behaviors,” a disconcerting term suggesting that residents could, and should, avoid acting in a disruptive way. A social worker in Texas who used to work in a nursing facility said the underlying issue is that “the nursing homes don’t want behaviors. They want docile.”
Many nursing home staff spoke to me about using antipsychotics to control residents as if it were a perfectly acceptable practice. Others told me that they had become aware that antipsychotic drugs were frequently misused only after the facility’s administration or corporate owners decided to cut down on their use. “It used to be like a death prison here,” a nursing director in a Kansas nursing home told me. “Half our residents were on antipsychotics. Only 10 percent of our residents have a mental illness.” Senior staff at the facility led an effort to reduce the antipsychotic drug rate after receiving financial penalties for administering unnecessary medications and after pressure from the chain’s owners. Another nurse in Kansas said: “We were at 55 percent antipsychotic drug rate before. Now we’re down to only people with a diagnosis [for which the FDA has approved the medications] on the drugs. They have a better quality of life because they’re not sedated.”
[I’m documenting my own Alzheimer’s disease while I still can]
Nursing homes, a mostly for-profit industry, control most aspects of their residents’ lives. Presumably, providers would be more inclined to meet minimum health and safety standards if it cost them dearly not to do so. “In this industry, there is a real cost-benefit analysis,” one long-term-care consultant told me. “If the fine will be $100,000, then they’ll hire the three nurse aides who will cost them about the same amount.”
Antipsychotic drugs may be an appropriate treatment for some people with dementia in nursing homes, but determining that requires a doctor and an informed patient (or proxy). And once told of the significant risks, unlikely benefits and possible alternative treatments — such as behavioral therapy, adjusting routine and environment, or alleviating the source of underlying loneliness, pain, boredom or fear — many people would probably reject the drugs. As one long-term-care pharmacist in Kansas said: “I don’t think antipsychotic drugs are presented well to the family in informed-consent conversations. Because if it were, they’d all reject it.”
An 81-year-old man in a Texas nursing home put it this way to me: “Too many times I’m given too many pills. I can’t even talk. I have a thick tongue when they do that. I ask them not to [give me the antipsychotic drugs]. When I say that, they threaten to remove me from the home. They get me so I can’t think. I don’t want anything to make me change the person I am.”
Hannah Flamm, an immigration lawyer at the Door’s Legal Services Center, reported on the misuse of antipsychotic drugs in nursing homes while she was a fellow at Human Rights Watch.