Summary
We lost colleagues. Others were intubated. It was really scary…. [I am] praying to have a vaccine soon.
—“Fernanda”, nurse at a public hospital in Ceará state, Brazil, July 28, 2020
Whoever finds the vaccine must share it. This is a global responsibility and it’s a moral responsibility for a vaccine to be shared far and wide. Some might see short-term advantage or even profit, but I assure you, to anyone who may think along those lines, humanity will have a very long memory, and be a very, very severe judge.
—Scott Morrison, prime minister, Australia, September 26, 2020, 75th United Nations General Assembly
The Covid-19 pandemic is among the gravest global health and economic crises in history. By mid-October 2020, it had taken the lives of more than a million and infected at least another 38 million, leaving many of them severely ill. Its social and economic consequences have been widespread and devastating. There is a growing movement of advocates, including Covid-19 survivors and loved ones of those who died, calling for a “people’s vaccine.”
World over, people are pinning their hopes on potential Covid-19 vaccines. The race to develop Covid-19 vaccines has made headlines nearly every day since the World Health Organization (WHO) first described Covid-19 as a pandemic in March 2020. By mid-October 2020, more than 40 vaccine candidates were in clinical trials, and more than 150 others were in earlier stages. Of the top 10 vaccine candidates in the final phase of clinical trials, companies or research institutes headquartered in China, Germany, Russia, the United Kingdom, and the United States were involved in developing them.
Universal and equitable access to a safe and effective Covid-19 vaccine is critical to ending the pandemic, or if no vaccine provides absolute immunity, preventing severe illness and death while protecting livelihoods and allowing battered economies to recover from the consequences of the pandemic.
Governments—especially high-income countries that can afford to do so—are using public money to fund Covid-19 vaccines on an unprecedented scale, with more than US$19 billion pledged by mid-September 2020. In mid-October 2020, the World Bank’s board of directors approved US$12 billion in financing for Covid-19 vaccines, testing, and treatment.
In the high stakes race for a potential vaccine, commitments to meet human rights obligations and transparency have been largely absent. Funding of Covid-19 vaccines with public money has been mired in secrecy. Rich governments that can afford to do so are negotiating opaque bilateral deals with pharmaceutical companies or other entities, often reserving future doses of vaccines largely for their exclusive use.
Secret deal-making and hoarding future vaccines in a climate where vaccines are widely projected to be in scarce supply—an approach often described as “vaccine nationalism”—have dealt massive blows to any global vision for universal and equitable access to an affordable vaccine and risks leaving people from low- and middle-income countries to pick through whatever is left after rich countries have served themselves. As Fatima Hassan, a South African human rights lawyer and intellectual property (IP) rights expert said, “How vaccines are distributed will expose the divides by race, class, and economic power.”
Bridging these divides in a way that respects the human rights of all is urgent and central to the world’s collective recovery from the pandemic. Human Rights Watch has carried out research examining the rights implications surrounding Covid-19 vaccine availability and affordability. Between April 23 and October 13, 2020, Human Rights Watch had discussions with more than 15 renowned experts on access to medicines, IP, and human rights; analyzed international human rights law and relevant national legal good practices; and reviewed a vast array of publicly available documents and secondary sources. This included epidemiological data; statements and resolutions by the WHO and the UN; reports or other documents by nongovernmental organizations (NGOs), pharmaceutical companies, multi-stakeholder initiatives, and others; media reports; and files from legal proceedings. Human Rights Watch also drew on more than six months of reporting on the pandemic’s impacts on different populations, including health workers.
This report examines key elements of a human rights-based approach to Covid-19 vaccines funding rooted in principles of transparency and accountability. It assesses how a variety of core rights—including but not limited to the rights to life, health, and an adequate standard of living—are being taken into account by governments. Human Rights Watch argues that governments spending public money on Covid-19 vaccines should take all possible measures within their power to ensure the scientific benefits of the research they fund are shared as widely as possible “to prevent the unacceptable prioritization of profit for some over benefit for all” and to maximize vaccine availability and affordability within the shortest possible time. While transparency has many aspects, this report looks closely at why governments should report transparently on their funding agreements, terms, and conditions. The concerns and recommendations Human Rights Watch raises in this report, particularly around barriers related to technology transfer and IP, and pricing, are relevant for other Covid-19 health technologies, including testing and drugs, but this report does not cover those areas.
“Sharing the Vaccine Recipe”
Right now, we are in the position where one corporation that is publicly funded may eventually discover or may eventually come across the vaccine recipe and that one corporation will not be able to supply the world. So it’s clear that there is a moral obligation on that corporation to share that recipe so all vaccine manufacturers in the world, so everyone who can, does start making the vaccine.
—Zain Rizvi, law and policy researcher, Public Citizen, July 24, 2020
High-income governments are using public money to preorder future vaccine doses in ways that undermine universal and equitable vaccine access. This has been with patent disregard for governments’ human rights obligations to cooperate internationally to share the benefits of scientific research they fund with public money to protect the rights to life, health, and a decent standard of living in a globalized world.
The UN Committee on Economic, Social and Cultural Rights, the body of experts that monitors implementation of the International Covenant on Economic, Social and Cultural Rights by its states parties stated in April 2020, “[P]andemics are a crucial example of the need for scientific international cooperation to face transnational threats … [i]f a pandemic develops, sharing the best scientific knowledge and its applications, especially in the medical field, becomes crucial to mitigate the impact of the disease and to expedite the discovery of effective treatments and vaccines…. The Committee reiterates that ultimately, intellectual property is a social product and has a social function and consequently, States parties have a duty to prevent unreasonably high costs for access to essential medicines.”
The funding amounts pledged and doses prebooked are frequently changing with new developments. The shifting environment is further complicated by a near total lack of transparency. Governments do not publicly disclose the terms and conditions of their funding, what they have prebooked and for whom, and which manufacturing facilities will be used to supply their preorders. NGOs, researchers, and journalists are piecing together this data.
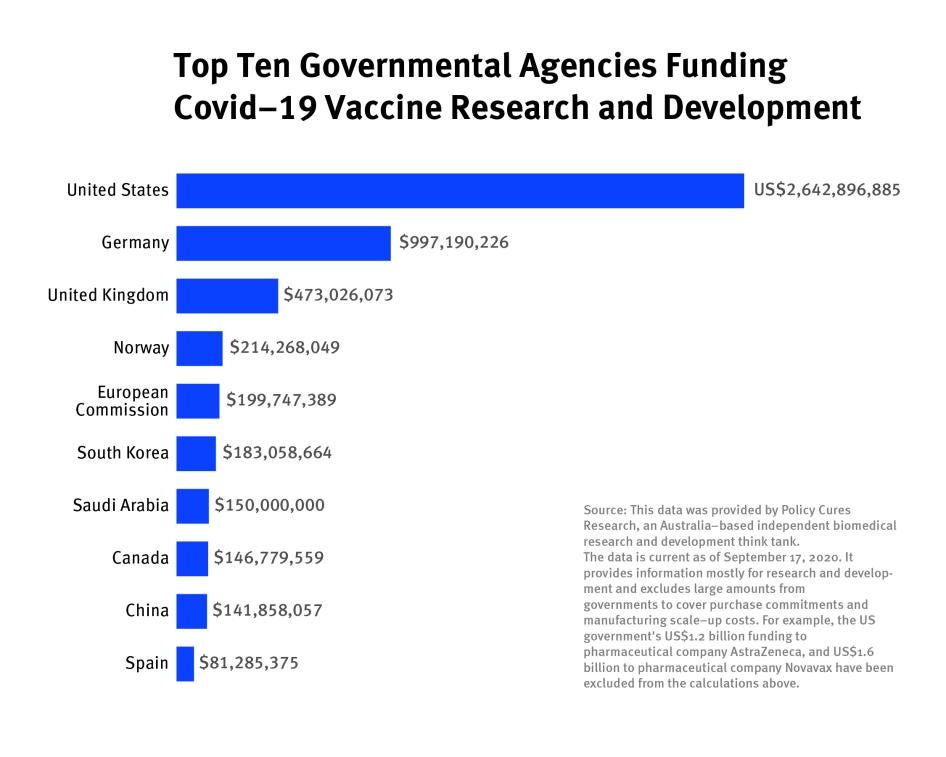
For example, Policy Cures Research, an Australia-based global health think tank, estimated that by mid-September 2020, the US, Germany, the UK, Norway, South Korea, Saudi Arabia, Canada, China, and Spain were the top nine governments, together with the European Commission, funding vaccine research and development, either directly through bilateral deals or indirectly by funding vaccine public-private partnerships like the Coalition for Epidemic Preparedness Innovations (CEPI). In September 2020, Oxfam International analyzed available information on deals made for five of the nine vaccine candidates that were in the final phase (“phase 3”) of clinical trials at that time. They estimated that 51 percent of the doses were reserved for high-income countries including Australia, Hong Kong & Macau, Israel, Japan, Switzerland, the UK, the US, and the European Union, even though they represent only 13 percent of the world’s population.
World leaders and experts have been clamoring for global cooperation and sharing of health technologies since the early days of the pandemic. Leading groups and experts on access to medicines who are following the funding and procurement efforts, including the European Public Health Alliance, I-MAK, Knowledge Ecology International, Médecins Sans Frontières (MSF) (Doctors without Borders), Oxfam, the People’s Vaccine Alliance, Public Citizen, South Centre, and the Third World Network, have repeatedly raised concerns that governments are not acting in ways that will ensure potential future Covid-19 vaccines will be a “global public good” accessible to all.
In May 2020, the Costa Rican government spearheaded a Solidarity Call to Action with the WHO to create the Covid-19 Technology Access Pool (C-TAP)—a common shared pool of rights to technologies, data, and know-how that everyone around the world could use to manufacture any medical products needed to tackle Covid-19, including vaccines. At time of writing, only 40 other governments had endorsed the C-TAP Solidarity Call to Action; most of them from low- or middle-income countries. The vast majority of high-income countries have yet to align with the principles and spirit of C-TAP.
Open and non-exclusive licensing unleashes the potential of scientific research that uses public money to maximize public benefit, preventing any one company from holding or controlling access to the data, know-how, and IP required to manufacture vaccines. Governments should use their regulatory powers to require companies and other entities to share data, know-how, and IP, including through open and non-exclusive licensing, to avert a scenario that could, as Fatima Hassan described it, allow drug companies to “play god” and “decide who gets a vaccine and when.”
This is especially important to expand global vaccine manufacturing capacity wherever possible, including in the long-term. As Third World Network, an NGO, has shown through its “VaxMap,” the expertise to manufacture vaccines is limited and highly skewed toward rich countries. Large parts of Africa, Asia, and Latin America do not have any significant manufacturing capacity and will likely depend on imports. Only a handful of countries in these parts of the world have vaccine manufacturing capacity: Argentina, China, India, Brazil, South Korea, and Singapore.
High-income governments have blithely ignored the repeated concerns raised by low- and middle-income countries about the significant barriers presented by IP, disregarding historical experience and lessons, especially from the struggle to overcome IP barriers for generic and affordable treatment for HIV/AIDS. This approach fails to learn lessons from past vaccine efforts and the links between pricing and IP, ignores expert opinions by IP rights lawyers, and disregards the growing risk of lawsuits challenging the IP of companies involved in Covid-19 vaccines. Voluntary corporate commitment to open and non-exclusive licensing has been low, making government use of regulatory tools essential to ensure vaccines are widely available.
In October 2020, India and South Africa proposed that some provisions of the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) be waived, to allow all countries globally to collaborate with one another, without running into the morass of laws and restrictions governing IP. If adopted, the proposal could enable the sharing of technology and know-how to expand access to the medical technologies needed to save lives and prevent severe illness during the pandemic. Human Rights Watch called on all other governments to support the proposal of India and South Africa.
The time to move beyond rhetoric around global cooperation is now. As James Love, director of the NGO Knowledge Ecology International, said: “It doesn’t take a genius to figure out that vaccines will be in short supply in the short-term. But why make it worse? Giving companies monopolies over vaccines in the middle of a pandemic is criminal.”
All governments should take concrete steps to endorse the C-TAP Solidarity Call to Action and develop a collective strategy to implement it. Governments should cooperate to urgently map out what vaccine manufacturing capacity exists, what has been booked up and for whom, and develop a plan of action to scale-up manufacturing to maximize the availability, affordability, and distribution of any successful vaccine. They should do so in consultation with low- and middle-income governments as well as experts on access to medicines, vaccines, IP, and human rights. Governments should also use their regulatory powers and funding clout to enable technology transfers and sharing of data, know-how, and other IP through open and non-exclusive licensing.
Making Vaccines Affordable for All
Thus far, some companies, universities, and institutes producing vaccines have given vague assurances of “non-profit” pricing and even provided pricing estimates. The estimates reported vary from as little as US$3 per dose to US$72 per dose. Most potential Covid-19 vaccines are expected to require two doses per person, and unlike some other vaccines, Covid-19 vaccines likely will not be once-in-a-lifetime. They will generate recurring expenses and be particularly financially burdensome for low- and middle-income governments as well as those living in poverty.
Regulating prices to maximize affordability is especially important because in many contexts, vaccines will only be accessible and affordable for communities if they are free at point of care. Worldwide, nine percent of the world’s population lives in extreme poverty, surviving on less than US$1.90 per day. In June 2020, the World Bank projected the pandemic could push between 70 and 100 million people into extreme poverty—living on less than US$1.90 a day—in 2020. The pandemic is also projected to widen the poverty gap between women and men and drive a 9.1 percent increase in poverty among women.
Governments have a human rights obligation to take measures, individually and collectively, to ensure companies, universities, and other entities receiving public money use it in ways that maximize vaccine affordability for the benefit of people globally. To this end, governments should use their powers to ensure vaccines are not sold to the highest bidder to maximize profits. Governments should require transparent pricing and third-party audits that demonstrate the company or university has maximized affordability and minimized debt for low- and middle-income countries.
|
Global Vaccine Procurement Mechanism: COVAX Facility There is growing interest from many governments—low-, middle-, and high-income—to purchase vaccines through a global procurement mechanism called the COVAX Facility. The COVAX Facility implemented by Gavi, the Vaccine Alliance—a public-private partnership—is operating subject to the preorder vaccine deals rich governments have already signed. The facility has yet to publish its contracts with companies and other entities. It has not endorsed C-TAP or taken steps to enable wide-ranging technology transfers or address IP barriers through open, non-exclusive licensing. Gavi estimates each vaccine dose supplied by COVAX will cost about US$10.55 and—for two vaccine doses per person—will total more than US$20 per person. Gavi has yet to require transparent pricing coupled with third-party audits to ensure companies producing vaccines for the COVAX Facility are maximizing affordability of vaccines. |
Committing to Transparency
People have a right to know how governments spend public money. They have a right to know what measures governments are taking to protect public health and ensure public money is used for public benefit. But governments have yet to disclose even the key terms and conditions under which they have funded companies, universities, and other entities for Covid-19 vaccines.
Transparency is crucial to hold governments accountable for fulfilling their international human rights obligations both within their own borders and beyond them. As Safura Abdool Karim, a senior researcher and health lawyer from the South African Medical Research Council’s Centre for Health Economics and Decision Science said, “Transparency is so pivotal…. We need to know what bilateral deals have been made, what are the preferential terms, and if there are any equitable access terms in these agreements. …Even the poorest of poor should be able to have access to a vaccine at an affordable price.”
The stakes have never been higher. Universal and equitable vaccine access is needed to save lives, protect health workers, prevent people from experiencing severe illness and potential long-term negative health impacts, ensure children return to school, and enable economies to rebuild and families to make ends meet. The lives and livelihoods of billions of people are on the line.
Key Recommendations
To All Governments
- Endorse and implement the WHO's COVID-19 Technology Access Pool (C-TAP).
- Collectively map out the global capacity to manufacture the needed supply of Covid-19 vaccines, and cooperate to develop a strategy to fund and support the creation of additional manufacturing capacity especially in low- and middle-income countries to ensure rapid and diversified large-scale production.
- Pledge not to sign bilateral deals with vaccine developers to prebook future vaccine doses in ways that undermine equitable global allocation according to public health need.
- Support India and South Africa’s October 2020 proposal at the World Trade Organization to waive some provisions of the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS Agreement) and use TRIPS flexibilities at the country-level.
To Governments Funding Covid-19 Vaccines
- Commit to transparency around Covid-19 vaccine funding by creating a public database of all contracts for funding related to Covid-19 vaccine research, development, manufacturing, or distribution.
- Attach concrete conditions to all funding for Covid-19 vaccine research, development, manufacturing, or distribution requiring technology transfer, that is, recipients should share all intellectual property (IP), data, and other know-how to enable mass manufacturing of successful vaccine candidates. Specifically, ensure agreements to fund Covid-19 vaccines, at a minimum, require recipients to provide open, non-exclusive, and royalty-free licensing for pre-existing IP owned and any future owned IP modeled on the “Open COVID Pledge.”
- Attach concrete conditions to all vaccine funding to ensure vaccines are priced transparently, and to prioritize affordability and minimize any debts for low- and middle-income countries and not profits for vaccine-developers. Specifically, ensure agreements to fund Covid-19 vaccines, at a minimum, require that compliance with vaccine price commitments is verified through an independent third-party financial audit and the audit report be published.
To Governments Participating in the COVAX Facility
- Call on Gavi and CEPI to endorse and design the COVAX Facility in a way that is aligned with the WHO’s C-TAP.
- Call on Gavi and CEPI to publish all agreements with vaccine developers and manufacturers.
- Call on Gavi and CEPI to attach conditions to their funding related to technology transfers; open and non-exclusive licensing; and transparent and affordable pricing, verifiable by independent third-party audits that are made public.
To UN Organizations
- Take measures to promote and facilitate technology transfers of any successful Covid-19 vaccine to make it as widely available as possible.
- Take measures to ensure any successful vaccine is priced affordably and made accessible to all vulnerable populations.
I. Introduction
Lives on the Line
“Fernanda” (not her real name), a nurse in her late 40s, cared for patients with Covid-19 in a government hospital in the state of Ceará, Brazil until April 2020, before authorities began redirecting patients to an isolation ward at another facility. Soon after, she tested positive for the novel coronavirus. “I have three kids, so I panicked,” she said. “How could I protect people at home? I feared that they would be infected as well.” Her husband and children experienced symptoms consistent with Covid-19 but were not able to get access to testing to confirm infection. “I’m still panicking today,” she told Human Rights Watch two months later, in mid-July 2020. “I have been extremely careful. We get very afraid of dying.” Fernanda feared more than half her co-workers had Covid-19, though not all were confirmed. “We lost colleagues. Others were intubated. It was really scary.” She said she is “praying to have a vaccine soon” as she felt, “we will only be safe with a vaccine.”[1]
More than 7,000 miles from Ceará, on the other side of the world, “Sister Noor” works in a ward exclusively for Covid-19 patients in a government hospital in Karachi, Pakistan. She told Human Rights Watch in May 2020, “We have direct and constant contact with patients all day long, whether we are putting in IV [intravenous] lines, or taking a blood sample, or checking their blood pressure, or giving them medicine.” She lamented the quality of the personal protective equipment (PPE) nurses received, adding, “Our caps will tear as we put them on, but doctors’ PPE gowns and caps are much sturdier.” She felt there was an informal pecking order for good quality PPE and wondered how and when vaccines—when available—would be distributed:
I fear the same thing will happen when the vaccines come—the doctors will get it first and we will be pushed to the back…. I don’t even want to think when the actual poor people in need will get vaccines. It will be hospital admin, doctors, politicians first, and everyone else later, if there’s any left.[2]
More than a million people around the world have lost their lives to Covid-19. At least another 38 million people have been infected, many with severe illness.[3] The total number of confirmed cases worldwide has grown exponentially since March 11, 2020 when the World Health Organization (WHO) first described Covid-19 as a pandemic. At that time there were 118,000 confirmed cases worldwide and nearly 4,300 deaths.[4] Seven months later, at time of writing, the world had more than 300 times more cases and more than 250 times more deaths, with the number of cases continuing to climb at an alarming rate. Many more cases and deaths have likely gone unreported due to low levels of testing and other factors.[5] Today, nearly every country in the world has reported Covid-19 cases.[6] The numbers alone cannot fully reflect the devastation of the pandemic on people around the world, especially older people and health workers.
Worldwide, millions of health workers like Fernanda and Sister Noor, about 70 percent of them women,[7] are risking their lives caring for Covid-19 patients. Underfunded and overwhelmed health systems, inadequate protective equipment, and little government support have exacerbated the risks they face. In September 2020, Amnesty International, the international human rights NGO, estimated at least 7,000 health workers worldwide had died of Covid-19.[8] Women health workers are more likely, as Sister Noor described, to be working in lower ranking roles, with less ability to demand safer working conditions.[9]
Many health workers spoke to Human Rights Watch about their experiences caring for Covid-19 patients. Adolfo Díaz Ruiz, a general surgeon in Nicaragua, said, “In the hospital where I work, at least 20 percent of the health workers have been infected at some point.”[10] A doctor in Soweto, South Africa said, “We are overstretched and are not sufficiently protected. Our wages are being reduced, but I still must pay for my own personal protective equipment (PPE). If I die, I am gone.”[11] In Venezuela, Bangladesh, Burundi, and Lebanon, health workers described precarious and often nightmarish working conditions and grave fear that they would lose their lives.[12]
The social and economic consequences of the pandemic have been widespread and far reaching. Among the pandemic’s many profound impacts are massive job losses and economic hardship with a disproportionate impact on women;[13] school closures disrupting the education of about 1.5 billion students;[14] spikes in reported cases of domestic violence in many locations;[15] and reduced access to basic health services, including routine immunizations[16] and sexual and reproductive health care, including abortion.[17]
The Race for Covid-19 Vaccines
Universal and equitable access to a safe and effective Covid-19 vaccine is critical to ending the pandemic, or if no vaccine provides absolute immunity, preventing severe illness and death while protecting livelihoods and allowing battered economies to recover.
The decisions governments make will determine how fast everyone—especially those in low- and middle-income countries—can have affordable access to a safe and effective vaccine, and whether it will be in time to protect their health and lives.
Pharmaceutical companies, universities, research institutes, multi-stakeholder initiatives, and other entities are racing to develop and manufacture safe and effective Covid-19 vaccines using a variety of different underlying technologies. Public health experts have consistently raised concerns about tremendous political pressure to develop successful vaccines.[18]
At time of writing, the WHO stated that 42 vaccine candidates were in human clinical trials.[19] In early September 2020, the Coalition for Epidemic Preparedness Innovations (CEPI), a global partnership between public, private, philanthropic, and civil society organizations that develops vaccines, reported the clinical trials planned to enroll at least 280,000 participants from at least 470 sites in 34 countries.[20] Many more vaccine candidates are in preclinical phases: the WHO tracked 156 such candidates,[21] while CEPI tracked many more in the exploratory phases.[22] Policy Cures Research, an Australia-based independent global health thinktank, had tracked funding for 203 potential vaccines as of October 1, 2020.[23]
Governments have invested an enormous amount of public money in vaccine research and development, and manufacturing and distribution, but as described in detail below, there is little or no information about the terms and conditions of their funding agreements.
Leading groups and experts on access to medicines who are following the funding and procurement efforts, including the European Public Health Alliance, I-MAK, Knowledge Ecology International, Médecins Sans Frontières (MSF) (Doctors without Borders), Oxfam, the People’s Vaccine Alliance, Public Citizen, South Centre, and the Third World Network, have repeatedly raised concerns that governments are not acting in ways that will ensure potential future Covid-19 vaccines will be a “global public good” accessible to all.[24]
The United Nations General Assembly in late April 2020 adopted a resolution urging the WHO, member states, and other stakeholders to “ensure fair, transparent, equitable, efficient and timely access to and distribution of” a range of tools to address Covid-19, including future vaccines, “with a view to making them available to all those in need, in particular in developing countries.”[25] In May 2020, 140 world leaders and experts in an open letter advocated that all Covid-19 diagnostics, treatments, and vaccines be treated as “public goods” and called for a “people’s vaccine” that is free of charge for those being vaccinated.[26] They wrote, “Access to vaccines and treatments as global public goods are in the interests of all humanity. We cannot afford for monopolies, crude competition and near-sighted nationalism to stand in the way.”[27]
On May 19, 2020, the World Health Assembly comprising of the WHO member governments, adopted a resolution that calls for “universal, timely and equitable access to, and fair distribution of, all quality, safe, efficacious and affordable essential health technologies and products, including their components and precursors, that are required in the response to the COVID-19 pandemic as a global priority,” including vaccines.[28] The resolution recognizes “the role of extensive immunization against COVID-19 as a global public good for health in preventing, containing and stopping transmission in order to bring the pandemic to an end, once safe, quality, efficacious, effective, accessible and affordable vaccines are available.”[29]
II. Vaccine Development and Funding Landscape
Key Global Developments concerning Vaccine Access
Since the World Health Organization (WHO) first characterized Covid-19 as a global pandemic in March 2020, there have been numerous efforts to encourage and facilitate greater global cooperation among governments and other stakeholders to ensure equitable access to a future Covid-19 vaccine and to fundraise for research, development, manufacturing, and delivery of safe and effective vaccines.
At the global level, there have been two approaches. The first is the WHO Covid-19 Technology Access Pool, or C-TAP, championed by the Costa Rican government, which promotes technology transfer and the global pooling and sharing of intellectual property (IP) and is largely aligned with the aims of the “people’s vaccine.”
The second approach is a vaccine procurement effort led by Gavi, the Vaccine Alliance, a “public-private partnership” that works to improve vaccine access.[30] Gavi’s “COVAX Facility,” through the WHO’s Access to COVID-19 Tools Accelerator (ACT Accelerator), had not required technology transfers or the open sharing of IP as conditions for companies receiving funds through the COVAX Facility at time of writing.
Covid-19 Technology Access Pool (C-TAP)
Following the May 2020 World Health Assembly resolution, the Costa Rican government in partnership with the WHO launched a Covid-19 Technology Access Pool (C-TAP), and issued a Solidarity Call to Action, which had been endorsed by 40 other governments at time of writing.[31] As the WHO says, C-TAP “will compile, in one place, pledges of commitment made under the Solidarity Call to Action to voluntarily share COVID-19 health technology related knowledge, intellectual property and data. The Pool will draw on relevant data from existing mechanisms, such as the Medicines Patent Pool and the UN Technology Bank-hosted Technology Access Partnership.”[32]
The Solidarity Call to Action urges all funders to take action to enable the sharing of knowledge, IP, and data for the Covid-19 response, and ensure all funding agreements help promote affordable and accessible Covid-19 related health products. It calls specifically for “global non-exclusive voluntary licensing, transparency and, when necessary, other commitments to expand access by sharing, for example, other intellectual property rights, know-how and data.”[33]
“ACT Accelerator” and the COVAX Pillar
In late April 2020, the WHO launched the Access to COVID-19 Tools Accelerator (ACT Accelerator), a collaborative platform for governments, major private funders, and scientists, calling on governments and private organizations to fund the “development and equitable distribution” of Covid-19 diagnostics, therapeutics, and vaccines.[34]
Two powerful multi-stakeholder initiatives—Gavi, the Vaccine Alliance, and the Coalition for Epidemic Preparedness Innovations (CEPI), a global alliance working to develop vaccines against infectious diseases—lead the Covid-19 vaccines pillar (“COVAX Pillar”) of the ACT Accelerator, together with the WHO.[35]
Gavi, CEPI, and the WHO identified a total funding need of more than US$18 billion for the development and delivery of up to two billion vaccine doses by the end of 2021.[36] Of the two billion doses, the COVAX Pillar plans to procure one billion doses for low- and middle-income countries by the end of 2021.[37] The roles of Gavi and CEPI in vaccine research, development, and procurement are analyzed further below.
WHO Equitable Allocation Framework
The WHO has developed a global, equitable, and fair allocation framework that seeks “to promote equitable access to COVID-19 vaccines, therapeutics and diagnostics that are in short supply” and “guide the process of allocation of COVID-19 products among countries (rather than within countries).”[38] In September 2020, the WHO published a separate document describing a “fair allocation mechanism for COVID-19 vaccines through Gavi’s COVAX Facility,” following the same principles outlined in the broader draft framework.[39]
Priority Target Groups
In both an August 2020 draft that was circulated to stakeholders and the September 2020 publication on vaccine allocation through the COVAX Facility, the WHO proposed that while the Covid-19 vaccine supply is scarce, countries should “focus initially on reducing mortality and protecting the health system,”[40] by identifying target groups based on current scientific and epidemiological data across diverse settings, and prioritizing within those target groups.
In the absence of any specific safe and effective vaccines, the WHO has proposed that “Tier 1,” or highest priority group for vaccination potentially include front-line health workers, people over age 65, and people with certain underlying conditions.[41] The WHO estimated that roughly 20 percent of a country’s population would cover most of the population comprising these groups.[42]
The WHO states that these three categories of populations have been identified assuming “a broad safety and effectiveness profile” of vaccines,[43] further stating that formal policy guidelines will be developed by the Strategic Advisory Group of Experts (SAGE) on Immunization—a body that advises the WHO—after “specific vaccines become available and are considered in terms of their safety and effectiveness in specific populations.”[44]
Phased Allocation
The WHO calls for all countries opting to participate in the COVAX Facility’s global vaccine procurement system to receive vaccines in two phases.[45] First, “an initial allocation of vaccines based on a proportional allocation scheme,” arguing that vaccinating 20 percent of each country’s population would likely “cover most of those in initially prioritized target groups to help to prevent numerous deaths, reduce the societal and economic consequences, and potentially change the course of the pandemic.”[46] The WHO states that “the fixed percentage allows for flexibility in the use of these doses according to national needs and contexts and according to the recommendations issued by SAGE once vaccines are available.”[47]
The WHO states that the initial vaccine supply will not be sufficient to cover 20 percent of the world’s population, and proposes each country be allocated a batch of vaccines to cover three percent its population, enabling—at a minimum—immunization of front-line health workers.[48] Then, additional batches of vaccines would be allocated proportionally until every country had received sufficient doses to cover 20 percent of its population.
Second, once 20 percent of each country’s population was vaccinated, subsequent allocation would be determined based on an assessment of epidemiological risk and the country’s vulnerability.[49]
Governments Funding Covid-19 Vaccines
Government agencies, especially from high-income countries,[50] are investing enormous amounts of public money in Covid-19 vaccine research and development, manufacture, and distribution. By mid-September 2020, this amounted to at least US$19.2 billion.[51]
Governments are pledging funds in different ways. Some governments, especially high- income countries, including the United Kingdom, the United States, Australia, Hong Kong and Macau, Japan, Switzerland and Israel, together with the European Union, are directly negotiating and funding bilateral agreements with pharmaceutical companies, universities, and research institutes to develop and manufacture vaccines, reserving future doses of vaccines.[52] Some governments are pledging funds to the “COVAX Facility,” a global vaccine procurement mechanism of the ACT Accelerator.[53] Some high-income countries are doing both, that is, signing bilateral agreements and reserving future doses of vaccines, and pledging to procure vaccines through the COVAX Facility.[54]
On October 13, 2020, the World Bank’s Board of Executive Directors approved a US$12 billion financing plan for low- and middle-income countries to fund Covid-19 vaccines, testing, and treatment.[55]
In addition to governments funding vaccines, many government agencies from a variety of countries have approved clinical trials for Covid-19 vaccine candidates in their countries. At time of writing, at least 34 countries were participating in clinical trials for various vaccine candidates.[56]
|
Vaccine Candidates Undergoing Phase 3 Clinical Trials (as of October 15, 2020)*
* Source: The WHO landscape of vaccine candidates, October 15, 2020 (latest available update at time of writing). |
The Role of Multi-Stakeholder Initiatives CEPI and Gavi
The Coalition for Epidemic Preparedness Innovations (CEPI) and Gavi, the Vaccine Alliance, play a critical role in vaccine access. CEPI and Gavi are global partnerships whose governance represents different stakeholders—public and private—including the pharmaceutical industry and corporate foundations. CEPI and Gavi have specific mandates to support low-income countries with vaccine access.[58] They receive funds from governments and other private sources, and in turn fund vaccine developers and manufacturers, respectively.
Gavi and CEPI lead the COVAX Pillar—the development, manufacturing, and distribution of vaccines as part of the Access to COVID-19 Tools Accelerator (ACT Accelerator) initiative, described above. Their roles are delineated: CEPI primarily disburses funds for vaccine research and development and at time of writing, eight vaccine candidates funded by CEPI were in clinical trials.[59] Gavi leads efforts to coordinate manufacturing and procurement through a global vaccine procurement system, the “COVAX Facility.”[60]
Gavi’s “COVAX Facility”: Global Vaccine Procurement
The COVAX Facility’s professed goal is to assist poorer countries that cannot sign bilateral deals to fund and procure vaccines, and to provide “an insurance policy” for high-income countries to pool their risk to avoid gambling on a particular vaccine candidate that may not turn out safe and effective, and reduce the chances of a Covid-19 “resurgence by ensuring that the rest of the world gets access to doses too.”[61]
It creates a procurement mechanism where countries can commit to purchasing a set number of vaccine doses through upfront financial contributions. The COVAX Facility will then use the money to make agreements with vaccine manufacturers, giving them market incentives to scale up production to meet the demand.[62]
In a September 2020 article, Gavi’s CEO explained the facility will work differently for rich and poor governments. Countries with the capacity to self-finance vaccine procurement (“self-financing” countries) will be allowed to request doses sufficient to vaccinate between 10 and 50 percent of their population, depending on the amount they pay.
Gavi has created two options for self-financing countries: a “Committed Purchase Arrangement,” or an “Optional Purchase Arrangement.” In the committed arrangement, self-financing countries agree to procure a specified volume of vaccine doses through the Facility, providing a lower upfront payment of only 15 percent of the total cost per dose, or US$1.60 per dose.
In the optional arrangement, self-financing countries will be required to pay greater upfront costs, but be allowed to “choose to opt out of receiving any [particular] vaccine, without jeopardizing their ability to receive their full share of doses of other candidates, subject to supply becoming available.”[63] According to Gavi, the optional purchase agreements would be more attractive to participating countries that have already reserved sufficient doses of a particular vaccine through bilateral agreements.[64]
A June 2020 report to Gavi’s board stated that where governments have already procured vaccines through bilateral deals, they “would be encouraged (but not required) to donate any doses they may not require to the Facility.”[65]
In addition to the self-financing option, Gavi also launched an Advance Market Commitment (AMC) for COVID-19 Vaccines, a mechanism designed to incentivize vaccine manufacturers to produce sufficient quantities of vaccines to supply 92 countries that are low-income, lower middle-income, or eligible for assistance from the International Development Association.[66] In late September 2020, Gavi’s board was expected to make additional decisions about “the level and extent of support to the AMC group, including co-financing or cost-sharing, operational support, and technical assistance.”[67] At time of writing, these decisions have yet to be made public.
Gavi states that the COVAX Facility will allocate vaccines to all participating countries at the same rate, proportional to their population, and further, that[68] “no country will receive enough doses to vaccinate more than 20% of its population until all countries in the financing group [emphasis added] have been offered this amount. The only exception is those countries who have opted to receive fewer than 20%.”[69]
In August 2020, Gavi stated that 80 countries that fell in the “self-financing” category expressed an interest in the COVAX Facility.[70] Taken together with the 92 AMC-eligible countries, the 172 countries represented more than 70 percent of the global population.[71] Gavi urged self-financing countries to make binding commitments to the facility by mid-September 2020 and to make their upfront payments by early October 2020.[72]
As of October 12, 2020, Gavi reported that at several dozen countries and economies had signed “commitment agreements,” and a number of others had expressed “non-binding” interest to participate.[73]
A broad range of civil society groups wrote to Gavi’s board of directors in June 2020 to express concerns about Gavi’s COVAX Facility and COVAX AMC and to urge Gavi to meaningfully involve civil society in its plans around the Facility.[74] In mid-August 2020, Gavi, CEPI, and the WHO issued a call for civil society and community organizations to participate in the Vaccines Pillar of the ACT Accelerator.[75]
The groups also made other recommendations to Gavi’s board of directors:
- allocate vaccines based on public health criteria for all countries;
- publish all agreements and contracts made with manufacturers, and demand transparency from pharmaceutical companies around their costs;
- pay no more than the at-cost price for future doses of Covid-19 vaccines;
- set clear conditions with transparent criteria for advance funds awarded to pharmaceutical companies, stipulating expectations if pharmaceutical companies do not deliver on vaccine manufacture or delivery;
- require manufacturers receiving its funds to share IP, technologies, know-how, and data in line with the principles of the WHO’s C-TAP;
- include non-government purchasers of vaccines in its deals; and
- be accountable to its partners and meaningfully involve civil society in its plans around the Facility.[76]
On September 29, 2020, the People’s Vaccine Alliance, a coalition of activists and organizations, wrote to the members of Gavi’s board seeking urgent changes to the COVAX Facility. The group urged Gavi’s board to endorse the WHO’s C-TAP and “use its huge market power to insist on sharing know-how, technology transfer and licensing intellectual property” to maximize vaccine production. The group also called for full transparency on pricing; a flat, affordable vaccine price for all countries participating in the COVAX Facility; and full civil society participation.[77]
III. Rights-Based Approach to Covid-19 Vaccines
It doesn’t take a genius to figure out that vaccines will be in short supply in the short-term. But why make it worse? Giving companies monopolies over vaccines in the middle of a pandemic is criminal.
—James Love, director, Knowledge Ecology International, New York, July 22, 2020
When deciding which population gets a vaccine first, public health considerations will clash with wealth considerations. How vaccines are distributed will expose the divides by race, class, and economic power…. Many clinical trials like those in South Africa are benefiting from public funding and countries are participating with the hope that they will get preferential access to vaccines… If we rely on the benevolence of the pharmaceutical industry, we’re not guaranteed access.
—Fatima Hassan, South African human rights lawyer and intellectual property rights expert, Cape Town, September 15, 2020
Like other infectious diseases, Covid-19 can spread rapidly across borders. Future vaccines may not provide lasting immunity, potentially leaving countries vulnerable to repeated cycles or waves of infection. The protection of any one country’s population from Covid-19 is highly dependent on and interconnected to the protection of populations in other countries. Protecting human rights in the context of a global pandemic requires the “international assistance and cooperation” that the International Covenant on Economic, Social and Cultural Rights (ICESCR) calls for.[78] The United Nations Committee on Economic, Social and Cultural Rights, which monitors compliance with the ICESCR underscored that “[p]andemics are a crucial example of the need for scientific international cooperation to face transnational threats.”[79] They concluded that “[i]f a pandemic develops, sharing the best scientific knowledge and its applications, especially in the medical field, becomes crucial to mitigate the impact of the disease and to expedite the discovery of effective treatments and vaccines.”[80]
Governments should do all they can to make safe and effective vaccines available to as many people as possible, as quickly as possible. The impact of inequitable distribution, especially where large portions of the world’s population, including vulnerable populations within individual countries, do not have timely access to a safe and effective vaccine when available, should not be underestimated. Universal and equitable vaccine access is needed to save lives, prevent people from experiencing severe illness and potential long-term negative health impacts, ensure children return to school, and enable economies to rebuild and families to make ends meet. The lives and livelihoods of billions of people are on the line.
The funding decisions governments make now will impact if universal and equitable vaccine coverage is achieved in a timely fashion. Governments and international actors including the UN and the World Health Organization (WHO) should act swiftly to ensure no entity has a monopoly over any potential vaccine. They should take all feasible measures toward that end, including ensuring no company, institute, or university can invoke intellectual property (IP) protections to create a monopoly, and any such entity using government or international funds to develop vaccines is required to share their technology and know-how to scale-up vaccine manufacturing as quickly as possible.[81] Governments should ensure global manufacturing capacity is expanded and used to the fullest possible extent to make vaccines. Governments should ensure vaccine doses are not sold to the highest bidder to maximize profits but are considered a public good to be pooled and distributed equitably, taking into account those with the most urgent needs.
Human Rights Watch analyzed international human rights law relevant to vaccine access, outlining crucial elements of a rights-based approach to Covid-19 funding and how rights obligations may be fulfilled by imposing specific conditions on funding to corporations, universities, research institutes, and other entities to maximize vaccine availability and affordability. Governments should take all available measures, including via regulatory provisions and funding terms and conditions, to ensure the benefits of scientific knowledge and its applications to respond to the pandemic are shared, and expand vaccine manufacturing and access. Global cooperation and transparency advance governments’ human rights obligations to take individual and collective steps, while “vaccine nationalism”—with governments racing to sign deals with developers reserving as many future doses of vaccines as possible for their exclusive use—does not.
Making Vaccines Widely Available
The huge need worldwide for a safe and effective vaccine demands preparation for large-scale production when one—or more—is identified. Vaccinating the world’s 7.67 billion people[82] would likely require at least double the number of vaccine doses (most vaccines require two doses per person for vaccination), which the world’s leading vaccine manufacturer predicts will happen only by 2024.[83]
Several barriers to scaling up the manufacture of Covid-19 vaccines could be addressed through government action, including conditions tied to vaccine funding that require technology transfer and the sharing of IP to allow mass manufacturing of successful vaccine candidates.
All governments should cooperate to urgently map out what vaccine manufacturing capacity exists and what has been booked up, and further scale-up manufacturing capacity, to maximize the availability, affordability, and distribution of any successful vaccine or vaccines. Such international cooperation is especially urgent because a vast majority of vaccines that are currently being developed have already been reserved by a handful of high-income countries.
Technology Transfer and Sharing of Intellectual Property
The key to universal access is to produce vaccine doses in vast numbers in multiple places. Pharmaceutical monopolies put up barriers that restrict production and drive up prices, putting profit ahead of public health. In these unprecedented times, pharmaceutical companies should pool their knowledge and governments should make this a condition of public funding. Our best chance of all staying safe is to have Covid-19 vaccines and treatments that are available for all—free from patents and other intellectual property barriers.
—Anna Marriott, health policy adviser, Oxfam, October 9, 2020
No corporation can supply the world … it’s going to take time for manufacturers to learn how to produce an eventual successful vaccine. So we should be working now on clearing the barriers and setting up a cooperative framework so qualified manufacturers everywhere can produce any successful vaccines. Right now, there’s just tremendous deference to the regime of confidentiality, corporate secrecy, proprietary exclusive rights, where one company will contract with another according to their commercial interests and working very hard to keep the technology under their control. I think that is very likely to lead to an extremely limited supply and long delays in getting to anything approaching global scale…. [W]e could cut those delays by years … if we could put ourselves on emergency footing.
—Peter Maybarduk, director, Access to Medicines Program, Public Citizen, July 24, 2020
Vaccine manufacturing is specialized and know-how is one of the key barriers to expanding production.[84] The WHO says transfer of technology is critical for manufacturing and distributing vaccines rapidly.[85] Technology transfer requires companies, universities, and institutes to share what they consider “proprietary” data and know-how about the vaccine and its manufacturing processes, including IP such as patents and trade secrets.
The UN Committee on Economic, Social and Cultural Rights, which oversees implementation of the ICESCR, recently reiterated “that ultimately, intellectual property is a social product and has a social function.”[86] The Committee called on states to “make every effort, in their national regulations and in international agreements on IP, to guarantee the social dimensions of IP, in accordance with the international human rights obligations they have undertaken” and warned, “a balance must be reached between intellectual property and the open access and sharing of scientific knowledge and its applications, especially those linked to the realization of other economic, social and cultural rights, such as the rights to health, education and food.”[87]
Governments should prioritize conditioning all funds, whether directly given to corporations, universities, or institutes, to require technology transfer and the open sharing of vaccine-related proprietary information. Without technology transfer and non-exclusive sharing of IP, vaccine manufacturing is likely to be highly limited and skewed toward rich countries. It will also significantly delay delivery of vaccine doses to low- and middle-income countries, further harming their public health and economies. Zain Rizvi, a law and policy researcher at Public Citizen, a nongovernmental organization (NGO), explained: “Technology transfer means sharing the vaccine recipe … so all vaccine manufacturers in the world, so everyone who can, does start making the vaccine.”[88]
Numerous governments have repeatedly raised alarms about vaccine access for low- and middle-income countries, specifically urging technology transfers and open sharing of IP. In May 2020, Costa Rica and other governments endorsed the Covid-19 Technology Access Pool.[89] In late June 2020, the African Union ministers of health and heads of delegation issued a communique highlighting the challenges for vaccine access in Africa. They called on all countries “to ensure that all relevant technologies, intellectual property, data and know how are openly and immediately made available and rapidly scaled-up.”[90] Without such technology transfer and sharing of relevant IP, experts say it will be impossible to build new vaccine manufacturing capacity where it does not exist.
These concerns come against the backdrop of rich governments signing bilateral deals and stockpiling future vaccines, undermining calls for global solidarity and cooperation toward equitable vaccine access. Leading public health groups and experts on access to medicine have repeatedly warned the current approach—shrouded in secrecy, competition, and a race to fund and conclude vaccine deals—has resulted in “vaccine nationalism” rather than cooperation.[91]
Suerie Moon, co-director of the Graduate Institute Geneva’s Global Health Centre, told Politico the scramble by rich countries to prebook vaccine doses “means that there will be very, very little vaccine available for the rest of the world and for countries that either don’t have the money or the production capacity within their own borders to access the vaccine.”[92]
Currently, there is no international effort to systematically map out the vaccine manufacturing capacity that exists and identify what has already been booked through various bilateral and other procurement efforts. NGOs and public health research organizations are piecing together this information with whatever data is published through various sources including government agencies, pharmaceutical companies, universities, and research institutes.
A September 2020 Oxfam International study revealed that rich countries with 13 percent of the world’s population had already prebooked more than half (51 percent) of the promised doses of several leading Covid-19 vaccine candidates.[93]
Oxfam analyzed available information on deals made around five of the nine vaccine candidates that were in phase 3 clinical trials in September 2020. Oxfam estimated that these five vaccine candidates could yield a total of 5.94 billion vaccine doses for the world—enough to vaccinate roughly 2.97 billion people worldwide, assuming each vaccine requires two doses. But supply deals negotiated with vaccine developers have already reserved nearly 90 percent (5.303 billion) of those doses. The majority (51 percent) are reserved for high-income countries including “the UK, US, Australia, Hong Kong & Macau, Japan, Switzerland and Israel, as well as the European Union,” even though these countries represent only 13 percent of the world’s population. Oxfam explained, “the remaining 2.575 billion doses have been bought by or promised to developing countries including India, Bangladesh, China, Brazil, Indonesia and Mexico among others,” and that 300 million doses of the AstraZeneca vaccine were pledged to the COVAX Facility.[94]
Edward Hammond with the NGO Third World Network explained the harm of governments signing bilateral deals and prebooking vaccines at this early stage: “These [bilateral government] contracts are potentially locking manufacturing into patterns that are harmful to the rest of the world, and developing countries in particular, by locking up production, and by contractually obligating the manufacture of vaccines that might not be very efficient or [potentially] less effective.”[95]
|
Case Study: Governments Prebooking Vaccines through Bilateral Deals with AstraZeneca[96] Several rich governments have signed deals with the UK-based biopharmaceutical company AstraZeneca to prebook millions of future Covid-19 vaccines doses. AstraZeneca is producing the vaccine developed by the UK’s Oxford University.[97] Among the deals that are known to exist (not a comprehensive list):
These governments have not disclosed specific details of their contracts or agreements. |
Where one company has signed agreements with numerous governments or procurement platforms like the COVAX Facility, there is no clarity on the order in which these doses will be disbursed. For example, in May 2020, French pharmaceutical company Sanofi, was heavily criticized when its CEO was widely reported as saying the US would be the first to get Sanofi’s vaccine because the US was the first to invest in its vaccine development.[102] A media furor and outrage by the French government led Sanofi to retract its comments.[103]
Low- and middle-income countries with very little domestic manufacturing capacity risk having to wait longer to get vaccine access. They cannot afford to enter into risky advance deals totaling billions of dollars with pharmaceutical manufacturers and therefore stand to lose out on equitable access. Gavi, CEPI, and the WHO set a goal of delivering two billion vaccine doses by the end of 2021, one billion of them for low- and middle-income countries.[104]
The Third World Network has developed an online map, “VaxMap,” that shows the “vastly unequal geographic distribution of manufacturing capacity of Covid19 vaccines.”[105] As VaxMap shows, large parts of Africa, Asia, and Latin America do not have any significant manufacturing capacity today and likely will be dependent on imports. Only a handful of countries in those regions of the world have vaccine manufacturing capacity: Argentina, China, India, Brazil, South Korea, and Singapore.[106]
Hammond, an expert on biologics manufacturing, and the creator of VaxMap, told Human Rights Watch that, in his assessment, “[o]utside of India, China, and South Korea … [there is] extremely limited capacity in developing countries. So that [global manufacturing] capacity is hugely geographically imbalanced.”[107]
So far, however, governments have not shown leadership and commitment to global cooperation. In a discussion with James Love and Thiru Balasubramaniam, IP rights experts with the NGO Knowledge Ecology International, Love summarized the problem: “The people funding vaccine R&D [research and development] with public money are doing it in ways that allow [vaccine developers] to monopolize [technological] know-how during a pandemic, a global emergency.” Emphasizing how the pandemic has put certain populations at risk, and decimated the economies of a lot of places, he said that such an approach, “has a massive economic impact in addition to the health impact. And it has human rights consequences.”[108]
|
Good Practice: Sharing Know-How for Covid-19 Antibody Treatment In mid-July 2020, Genentech Inc. and other biotechnology companies received permission from the US Department of Defense to share their know-how around manufacture of a potential antibody treatment for Covid-19 currently in late-stage clinical trials. If the treatment is approved by the US Food and Drug Administration (FDA), the companies argued that sharing their know-how could speed up production of the treatment significantly.[109] The agreement is an example of the kind of collaboration public and private actors should take around vaccine development and manufacture. |
A legal battle involving Inovio Pharmaceuticals’ vaccine candidate demonstrates how IP rights claims and legal battles can become an impediment to rapid manufacturing.
|
Mired in Court Cases: Inovio Pharmaceuticals’ Covid-19 Vaccine Candidate[110] Inovio Pharmaceuticals, Inc., an American biopharmaceutical company, is funded by numerous donors, including US government agencies and CEPI, to develop and manufacture its Covid-19 vaccine candidate, INO-4800.[111] Inovio’s vaccine candidate is being developed using DNA technology. Its goal is to produce one million doses of the vaccine by the end of 2020 and further expand production.[112] But its vaccine development efforts are the subject of a legal battle. In June 2020, Inovio initiated legal proceedings against its manufacturing partner, VGXI (American subsidiary of South Korean GeneOne Life Science Inc) for breach of contract because it wants VGXI to transfer manufacturing technology to other manufacturers to speed up production goals it claims VGXI cannot meet in a timely fashion.[113] Inovio claims: “VGXI has repeatedly told Inovio, orally and in writing, that it cannot manufacture further batches of Inovio’s vaccine at any time in 2020 because it does not have manufacturing capacity to do so.”[114] According to Inovio, VGXI is building a new manufacturing facility that may be available by early 2022, “[b]ut in this environment, that is at least a year or two way [sic], and the world simply cannot wait for VGXI because every minute counts.”[115] Inovio further claims: VGXI’s breach is causing irreparable harm, and will continue to do so because its failure to transfer the technology is delaying the scale-up, manufacture, and availability of the vaccine, as Inovio and other manufacturers will need to start over and develop an FDA [Federal Drugs Authority]-approved process of manufacturing the vaccine…. If VGXI refuses to permit Transfer Technology, Inovio cannot manufacture the product promptly, safely, or perhaps at all.[116] Inovio contracted two other manufacturers, German company Richter-Helm and US company Ology Bioservices to scale up manufacturing.[117] Inovio claims both manufacturers are dependent on VGXI transferring manufacturing technology to start production at the earliest.[118] GeneOne Life Science countersued Inovio and its US manufacturing partner, Ology Bioservices in July 2020.[119] GeneOne claims in its lawsuit: “Inovio got greedy: it saw the opportunity to reap vast riches and keep its stock price soaring” by, GeneOne Life Science claims, “secretly planning for months to take VGXI’s intellectual property and disseminate it to other manufacturers.”[120] The dispute is ongoing. |
The Inovio case also shows the disputes that can arise when not all the IP surrounding the actual manufacture of the vaccine is owned by those developing it.
The patent landscape for vaccines is complex. A 2011 study by the WHO, the World Trade Organization (WTO), and the World Intellectual Property Rights Organization (WIPO) states that a “modern vaccine is protected by multiple levels of IP often licensed from multiple partners.”[121]
The multiple layers of IP owned and licensed are significant barriers to scaling up and rapidly manufacturing Covid-19 vaccines. Dr. Leena Contarino and Ellie Purnell, IP experts from HGF, a specialist global IP law firm, wrote:
Calls to make all Covid-19 vaccines freely available have been widespread and in some cases companies have already agreed either not to file patents on the novel treatments at all, or to provide royalty free licences to the patents they do file. However, the situation is more complicated than simply allowing or agreeing access to the patent/s protecting the vaccines. In many instances, the new Covid-19 vaccines will be built on proprietary platform technologies, such as viral vectors, mRNA-technology, nanoparticles and the like, which in themselves may be covered by multiple earlier patents owned by multiple different parties. Access to these platform technologies is also required if a novel vaccine incorporating the technology is to be produced and sold.[122]
The number of patents implicated is unknown but likely large. For instance, a 2012 WIPO, WHO, and WTO study tracking patents for vaccines related to infectious diseases found 11,818 families of patents and patent applications (a patent family is a set of patent filings made in various countries to protect a single invention) filed between 1921 and 2011.[123] Most of them were first filed in the US. Other top patent “first filing” destinations included China, the UK, Japan, Russia, the European Patent Office, France, Australia, South Korea, Germany, Denmark, Canada, Spain, Brazil, Sweden, India, Ukraine, Netherlands, and Cuba.[124]
The Need for Contractual Curbs on Exclusive Licensing
Exclusive licensing—when one party grants exclusive rights to another for the use of a specific patent or piece of IP—can be a serious barrier to equitable access and can exacerbate vaccine shortages by limiting the generic scale-up of production. Government contracts for Covid-19 vaccines should use open and non-exclusive licensing, allowing any qualified party to make use of the technology. For example, even though the UK government’s contract with Oxford University to develop a vaccine is yet to be published, it appears the government did not contractually curb Oxford University’s power to negotiate exclusive global licenses. Oxford University then signed an exclusive global license with UK pharmaceutical company, AstraZeneca, to use its technology to manufacture vaccines.[125]
The exclusive license drew widespread criticism from access to medicines experts. For example, Roxana Bonnell, senior program adviser at the Open Society Foundations’ Public Health Program and access to medicines expert, argued exclusive licensing hinders rapid manufacturing: “Exclusive licensing promotes profits, not public health, leaving a single corporation to decide where a vaccine is available and at what price. With a global pandemic like Covid-19, we need the opposite—global, decentralised manufacturing enabled by non-exclusive licensing, so that vaccines can be produced at the volumes needed.”[126]
Where a company does not voluntarily provide open and non-exclusive license for the use of its IP, it limits others’ ability to manufacture more vaccine doses. Various companies and universities, including Facebook, Amazon, Intel, Microsoft, HP have voluntarily adopted the “Open COVID Pledge,” pledging to make their “intellectual property available free of charge for use in ending the COVID-19 pandemic and minimizing the impact of the disease.”[127] The pledge is not focused on Covid-19 medical products, including vaccines. While those that have adopted the pledge are predominantly electronic companies, it also includes a few companies that have adopted the pledge in relation to IP for medical products.[128]
Companies taking the pledge issue an Open COVID License, which grants a “non-exclusive, royalty-free, worldwide, fully paid-up license (without the right to sublicense) under Pledgor’s patents to make, have made, use, sell, and import any patented invention, solely for the purpose of diagnosing, preventing, containing, and treating COVID-19.”[129]
Companies can choose the standard-format license or publish an alternative license. All standard-format Open COVID Licenses are effective as of December 1, 2019 regardless of when it was first granted. The licenses will last until January 1, 2023 and can be further extended by the pledgor.
While the “Open COVID Pledge” is an important initiative, none of the big pharmaceutical companies, universities, and institutes that are currently involved in Covid-19 vaccine development or manufacture have joined,[130] illustrating the limits of relying on voluntary commitments to open and non-exclusive licensing.
Making Vaccines Affordable
Governments have obligations under international human rights law to respect, protect, and fulfill the rights to life, to health, to an adequate standard of living, and to enjoy the benefits of scientific progress, among others.[131] For a state party to the ICESCR these obligations are to be met by taking steps “to the maximum of its available resources.”[132] Governments directing money into vaccine development and manufacture from their available resources should therefore be imposing conditions on that funding to ensure corporations and others eventually selling vaccines price them in a way that maximizes affordability and equitable access, and minimizes debt for low- and middle-income countries.
Pricing directly influences how many vaccine doses governments can buy and the extent to which they can make them freely available for populations in their countries. Past vaccine efforts—for example, for human papilloma virus (HPV) vaccines to prevent cervical cancer and pneumococcal vaccine (PCV) for children to prevent pneumonia-deaths in low- and middle- income countries—have repeatedly shown that IP rights have a direct bearing on vaccine pricing and in turn, vaccination efforts.[133]
Governments have obligations to ensure health facilities, goods, and services are accessible to everyone without discrimination, and affordable for all. These obligations are owed primarily to those within each government’s jurisdiction, but depending on resources, states also have obligations to help other governments realize those rights in their countries.[134]
Worldwide, nine percent of the world’s population lives in extreme poverty, surviving on less than US$1.90 per day. In April 2020, the World Bank warned, due to the Covid-19 crisis and the drop in oil prices, “For the first time since 1998, poverty rates will go up as the global economy falls into recession and there is a sharp drop in GDP per capita.”[135] The pandemic is also projected to widen the poverty gap between women and men and drive a 9.1 percent increase in poverty among women.[136] In June 2020, the World Bank projected the pandemic could push between 70 and 100 million people into extreme poverty—living on less than US$1.90 a day—in 2020.[137] People living in poverty are more likely to live in crowded or poor-quality housing, and lack the resources to stay at home for long periods or follow hygiene or social distancing recommendations. Low-paid jobs with poor working conditions force them to choose between risking their health or losing their income.[138]
This means in many contexts, vaccines will only be accessible and affordable for communities if they are free at point of care. Governments should attach conditions to their funding agreements to ensure vaccines will be priced to enable governments or international organizations to procure them at an affordable rate that does not jeopardize their ability to meet their other social and economic rights obligations. International cooperation should be directed towards supporting governments to ensure they can distribute vaccines free of cost to populations that cannot afford to pay.
Some recipients of government funding—universities and pharmaceutical companies— have suggested they will adopt pricing strategies that are “not-for-profit” or at minimal profit.[139] The information thus far disclosed is scant or vague. Where a little more information about potential pricing is made available, experts have raised concerns that the cost risks putting vaccines out of the reach of poor countries and people living in poverty. For example, in July 2020, Priti Krishtel, the co-founder of I-MAK, responded to early pricing information that was publicly available at that time:
When we see the first prices that are being announced, the only public prices that we really have to date, are saying about [US]$10-13 per dose. Now if that’s true, that’s more than twice what we are seeing being paid for the most expensive vaccine in the poorest countries.[140]
Pricing information that has become available over time shows prices can vary hugely, and also depend on the underlying technological platform used. Pricing in the case of Covid-19 vaccines is particularly important because these may not be once-in-a-lifetime vaccines, therefore creating recurring expenses that could be a barrier to access for low- and middle-income governments as well as people living in poverty.[141]
The US government in mid-July 2020 signed a US$2 billion deal with pharmaceutical giant Pfizer to reserve 100 million doses of a Covid-19 vaccine the company is testing.[142] The government agreed to pay Pfizer’s price of approximately US$20 per dose for the vaccine, which may involve a two-dose regimen costing about US$40 per person.[143] Another news outlet reported that a vaccine being developed by Sinopharm, a Chinese company, would cost about US$144 for two doses.[144] The lowest estimate thus far appears to have been projected by the Serum Institute of India, which announced it would manufacture AstraZeneca/Oxford and Novavax vaccines at a price capped at US$3 per dose for low- and middle-income countries, costing US$6 per person for a two-dose vaccine course.[145]
Gavi claims the COVAX Facility will “use the collective purchasing power that comes from having so many countries participate in order to negotiate highly competitive prices from manufacturers that are then passed on to participants.”[146] In a September 2020 document, Gavi stated the COVAX Facility will try to negotiate prices that are “lower than or at least no higher than pricing manufacturers have agreed in bilateral deals”—which vary widely, as Human Rights Watch explained above. Gavi has made an “estimated all-inclusive price per dose of [US]$10.55.”[147] Gavi has made clear that deals between manufacturers and the facility “will dictate the final price of the vaccine and whether the pricing structure is flat/single or tiered.”[148] At time of writing, Gavi was yet to require transparent pricing and third party audits.
Urgent Need for Governments to Invoke their Regulatory Powers
Governments should use their regulatory powers to maximize the manufacturing capacity for, and the affordability of, Covid-19 related medical products, including vaccines. This includes pricing regulations as well as regulations for compulsory licensing covering all Covid-19 medical products, including vaccines.
Using Compulsory Licenses to Scale up Manufacturing
Under the WTO Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) governments can issue compulsory licenses, that is, give third parties the right to use a patent without the authorization of the patent holder. “Compulsory license” is used to describe different types of non-voluntary licenses that a government can issue.[149] In some situations, for example, where the government itself seeks to use the patent for “public non-commercial use,” it does not have to seek prior authorization of the patent holder. Similarly, governments can dispense with the need to seek a voluntary license before issuing a compulsory license during emergencies.[150]
The 2001 Doha Declaration on the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) (the Doha Declaration) reiterated that IP rights should not be an impediment to protecting public health.[151] The Doha Declaration was negotiated in the midst of efforts to expand access to affordable generic medicines to treat HIV/AIDS. At the time, 9,000 people a day were dying of AIDS, largely in countries where antiretroviral drugs were unavailable or unaffordable.[152] It clarified that in case of public health concerns, including during epidemics, governments have the right to issue compulsory licenses, that is, permitting its use for generic manufacture without the permission of IP holders.[153] The WTO lifted limitations on exports of products manufactured under compulsory licenses to least-developed countries and other countries that have insufficient or zero manufacturing capacities in the pharmaceutical sector.[154]
Trade and IP rights experts have raised concerns about the legal amendments needed at national levels to give effect to TRIPS flexibilities and overly bureaucratic processes that exist at national levels, rendering the issuance of compulsory licenses complicated and time-consuming. A 2019 study by WIPO found that at least 156 countries and territories allow compulsory licensing under their respective legal frameworks.[155] To varying degrees the domestic procedural requirements for issuing licenses create significant time-consuming barriers.[156] For example, some countries’ laws require those applying for compulsory licenses to demonstrate they have “waited” for a “reasonable period” for a voluntary license after requesting one, and sometimes include waiting periods of 180 to 210 days.[157] Licenses can also be hindered by challenges brought through judicial review or other litigation.[158]
In July this year, South Africa wrote to the TRIPS Council, a body tasked with administering the treaty, raising concerns about the “legal, technical and institutional challenges in using TRIPS flexibilities,”[159] pointing out that many countries’ patent laws either do not allow for compulsory licenses or involve time-consuming processes.[160] Similar concerns were also echoed by Hu Yuan Qiong, the senior legal and policy adviser for the MSF Access Campaign.[161]
Further, 37 governments—Australia, Canada, Iceland, Japan, New Zealand, Norway, Switzerland, the UK, the US and the 27 EU member states—have opted out of importing products manufactured using compulsory licenses under TRIPS.[162] This makes it harder for manufacturers to produce cheaper products through economies of scale, thereby undercutting the economic viability of manufacturers who produce goods under compulsory licenses. A broad range of civil society groups, including Knowledge Ecology International, MSF, and Public Citizen, have called on these governments to reverse their decision to opt out of receiving generic imports.[163]
At time of writing, the Indian and South African governments had submitted a proposal to the TRIPS Council recommending a waiver from some provisions of the TRIPS Agreement “until widespread vaccination is in place globally, and the majority of the world’s population has developed immunity.”[164] If adopted, the proposal could enable technology transfer and widespread sharing of IP to expand access to testing, treatment, and potential vaccines needed to save lives and prevent severe illness during the Covid-19 pandemic.
|
Good Practices Around Compulsory Licensing Since the Covid-19 pandemic began, several governments have introduced new compulsory licensing regulations.[165] According to an August 2020 report by Public Citizen, “Germany, France, Canada and Indonesia have adopted new non-voluntary patent licensing policies. Several have taken steps to license patents, with Chile and Ecuador calling for licenses and Israel issuing a license for a candidate COVID-19 treatment.”[166] In the past, there have been numerous instances where governments issued compulsory licenses, including for government-use, for public health, especially for HIV/AIDS treatment. According to Ellen ’t Hoen, public health expert and founder of Medicines Patent Pool, between 2001 and 2014, at least 35 governments issued about 50 government-use licenses, mostly for specific drugs to treat HIV/AIDS.[167] In 2001, the US government threatened the pharmaceutical company Bayer with a compulsory license on ciprofloxacin (Cipro), which the government was stockpiling after letters containing anthrax spores were sent to media companies and congressional offices.[168] Bayer responded by lowering the cost of the drug significantly.[169] |
IV. Why Transparency Matters
Transparency is so pivotal…. We need to know what bilateral deals have been made, what are the preferential terms, and if there are any equitable access terms in these agreements.… Even the poorest of poor should be able to have access to a vaccine at an affordable price.
—Safura Abdool Karim, senior researcher and health lawyer, South African Medical Research Council’s Centre for Health Economics and Decision Science, Johannesburg, September 9, 2020
My concern is around the lack of transparency around the main agreements…. We can’t rely on investigative journalists and civil society speaking to people off the record to find out what’s going on, who put what money where and under what terms and conditions… It’s very difficult to actually know what is going on.
—Fatima Hassan, South African human rights lawyer and intellectual property (IP) rights expert, Cape Town, September 15, 2020
Governments funding vaccine research, development, and manufacture should publish contracts they sign with corporations, institutes, universities, and other entities. Transparency about what has been funded and the terms and conditions are critical for not just government accountability but also holding recipients to account. To date, governments have made very little information publicly available around their funding agreements. Governments should commit to transparency by publishing contracts and reporting in detail on terms, conditions, and other aspects of funding for Covid-19 vaccines.
Governments should also collaborate and publish all information they have about recipients’ pre-existing patents and licenses that are relevant to vaccine development and manufacture. This will help cut through the time needed for all parties to procure voluntary and compulsory licenses.
Transparency around contracts can also expose lax government oversight and contractual “deregulation.” Currently, there is little or no public information about whether governments funding vaccines have included “march-in” rights, that is, the right to override any exclusive licensing arrangements. The little information trickling out through freedom of information applications in the US shows a dismal trend. For example, analysis by Knowledge Ecology International (KEI), a nongovernmental organization with expertise on IP, showed that the Biomedical Advanced Research and Development Authority (BARDA) and the Department of Defense (DoD) had effectively watered down their statutory regulatory oversight. KEI received heavily redacted copies of seven agreements BARDA and DoD had signed with companies for Covid-19 vaccines and therapeutics in response to Freedom of Information Act (FOIA) requests they filed.[170]
KEI’s analysis of these agreements found the government had contractually limited or eliminated the government’s rights in inventions and data that was publicly funded. It also found the contracts had renegotiated terms, watering down statutory definitions, and had effectively undercut the powers of US government agencies under the Federal Acquisition Regulation (legislation governing executive acquisitions) and the US Bayh-Dole Act (legislation governing IP rights to federally funded inventions). The Bayh-Dole Act establishes mechanisms for US government agencies to assert certain claims over inventions that are funded by the federal government. This includes “march-in” rights as well as rights of the government to intervene if the invention’s practical application was not made available on “reasonable terms.” KEI found BARDA and DoD had signed contracts with five companies that had “limited its ability to intervene if the pharmaceutical companies (which are party to the agreements and are receiving hundreds of millions of dollars to conduct the research) charge unreasonable prices for the resulting COVID-19 vaccines or treatments.”[171]
V. International Legal Framework
Governments’ international human rights obligations include obligations to respect, protect, and fulfill the rights to life, to health, to an adequate standard of living, and to enjoy the benefits of scientific progress.[172] In the context of the Covid-19 pandemic, the vaccine nationalism and hoarding described above create barriers for many governments seeking to fulfil their human rights obligations, particularly regarding the right to health. Instead, governments should place not only the rights to life and health at the center of their response, but their obligations concerning international cooperation and extraterritorial impacts.
States’ Extraterritorial and International Cooperation Obligations, in the Context of the Right to Health
The obligation of international cooperation has multiple sources. The UN Committee on Economic Social and Cultural Rights (CESCR) has repeatedly emphasized that states parties to the International Covenant on Economic, Social and Cultural Rights (ICESCR)—of which there are 171—should recognize the essential role of international cooperation and comply with their commitment to take joint and separate action to achieve the full realization of economic and social rights.[173] The duty for UN members “to take joint and separate action in cooperation with the Organization” to achieve “universal respect for, and observance of, human rights and fundamental freedoms for all without distinction as to race, sex, language, or religion,” set out in articles 55 and 56 of the UN Charter, is expressed without any territorial limitation and is to be taken into account when addressing the scope of states’ obligations under human rights treaties.[174] Article 28 of the Universal Declaration of Human Rights (UDHR) also sets out that everyone is entitled to a social and international order in which the rights and freedoms in the UDHR can be fully realized, setting an expectation for states to contribute to creating an enabling international environment for the fulfilment of economic and social rights everywhere.[175] In particular, article 2(1) of the ICESCR creates an obligation on states to take collective action, including through international cooperation, to help fulfil the economic, social, and cultural rights of persons outside of their national territories.[176]
International cooperation requires states to refrain from actions that interfere, directly or indirectly, with the enjoyment of rights in other countries; no state should act in ways that frustrate the efforts of other states to comply with their human rights obligations, including when negotiating international agreements or participating in decisions as members of international organizations.[177] Activities undertaken within one state’s jurisdiction should not deprive another state of the ability to realize the rights for persons in its jurisdiction. On the contrary, the CESCR has held that depending on the availability of resources, states should facilitate realization of economic and social rights in other countries, for example through provision of resources, financial and technical assistance, and providing aid when required.[178]
States are also required to “direct the efforts of business entities” towards the fulfilment of the rights set out in the ICESCR and to that end have specific extraterritorial obligations concerning business activities.[179] States should control the activities of corporations under their jurisdiction, so that they do not interfere with, but rather might contribute to the effective enjoyment of economic, social, and cultural rights outside their national territory.[180] At a minimum, states should encourage business actors whose conduct they are in a position to influence not to undermine the efforts of the states in which they operate to fully realize economic and social rights.[181] The CESCR has emphasized that this duty is particularly relevant to the negotiation and conclusion of trade and investment agreements.[182]
Article 12 of the ICESCR provides that everyone has the right to “the highest attainable standard of physical and mental health.”[183] States are required to take steps “individually and through international assistance and cooperation, especially economic and technical, to the maximum of its available resources” to achieve the full realization of the right to health.[184]
To realize this right, one of the priority obligations of states is to take effective steps for the “prevention, treatment and control of epidemic, endemic, occupational and other diseases.”[185] The control of epidemics requires, among other steps, “the implementation or enhancement of immunization programmes and other strategies of infectious disease control.”[186] The CESCR in its authoritative guidance to states on the right to health states that “for the avoidance of any doubt … it is particularly incumbent on States parties and other actors in a position to assist, to provide “international assistance and cooperation, especially economic and technical” to enable developing countries to fulfil this obligation.[187]
The committee explained, “[g]iven that some diseases are easily transmissible beyond the frontiers of a State, the international community has a collective responsibility to address this problem. The economically developed States parties have a special responsibility and interest to assist the poorer developing States in this regard.”[188]
The right to health facilities, goods, and services includes “the provision of equal and timely access to basic preventive” services, “appropriate treatment of prevalent diseases,” and “the provision of essential drugs.”[189] States have an immediate obligation to “provide immunization against the major infectious diseases occurring in the community.”[190]
States must ensure health services are economically accessible, meaning “affordable for all” and of good quality.[191] In addition, “States have a special obligation to provide those who do not have sufficient means with the necessary health insurance and health-care facilities. …For example, investments should not disproportionately favour expensive curative health services which are often accessible only to a small, privileged fraction of the population, rather than primary and preventive health care benefiting a far larger part of the population.”[192]
Regarding international assistance, the CESCR has stated that, depending on the availability of resources, states should facilitate access to essential health facilities, goods and services in other countries, wherever possible and provide necessary aid when required. When concluding international agreements, states should take steps to ensure these instruments do not adversely impact upon the right to health and states have an obligation to ensure their actions as members of international organizations take due account of the right to health.[193]
Finally, “when designing a framework on intellectual property rights, consistent with the Universal Declaration of Human Rights and with the right to enjoy the benefits of scientific progress stipulated in article 15” of the ICESCR, the CESCR has warned states they should ensure intellectual property (IP) rights do not lead to denial or restriction of everyone’s access to essential medicines necessary for the enjoyment of the right to health.[194] The Committee has pointed out that states have a positive duty “to make available and accessible to all persons, without discrimination, especially to the most vulnerable, all the best available applications of scientific progress necessary to enjoy the highest attainable standard of heath.”[195]
In 2009, a gathering of experts from United Nations bodies including the Office of the High Commissioner for Human Rights, the CESCR, the World Intellectual Property Rights Organization (WIPO), and the World Trade Organization (WTO), led by UNESCO and including other experts from academia issued the Venice Statement on the Right to Enjoy the Benefits of Scientific Progress and its Applications.[196] The Venice Statement noted, “advances in medicine have helped to cure more diseases and enhance the quality of life. However, these advances are driven primarily by market considerations that often do not correspond to the health needs of the world’s population as a whole, thus affecting the right to health.”[197] It went on to conclude “the intellectual property regime, which is a temporary monopoly with a valuable social function that should be managed in accordance with a common responsibility to prevent the unacceptable prioritization of profit for some over benefit for all.[198]
In the context of a pandemic and in light of the requirement that states do not permit IP protections to impede realization of other economic, social, and cultural rights, such as the rights to health, this obligation should be interpreted to require states to ensure IP and licensing restrictions do not create a barrier to access health care.[199]
VI. The Way Forward
The tendency to protect national interests and prioritize profit and financial interests over people’s lives … is not going to disappear. This is why we need to establish a rights-based framework that articulates why and how Covid-19 vaccines, and other medical tools to curb the pandemic, must be seen as global public goods and be made available to all people in equitable ways.
—Fatima Hassan, South African human rights lawyer and intellectual property rights expert, and Els Torreele, access to medicine researcher and former executive director of the MSF Access Campaign, July 2020[200]
The Covid-19 pandemic has shown that the fates of people all over the world are interconnected: protecting one country’s people and its economy from the impacts a deadly infectious disease is impossible unless the people of other countries are also protected. Governments have a critical role to play in funding efforts to develop safe and effective vaccines. But no amount of funding will guarantee equitable access without decisive collective action and global cooperation to challenge the profit-driven and opaque systems that have determined access to lifesaving treatments and vaccines in the past.
Governments should continue to fund Covid-19 vaccines, especially to ensure access for low- and middle-income countries. While doing so, they should take all possible measures, including directing and conditioning funds in ways that are aligned with their human rights obligations to share the benefits of scientific knowledge and its applications widely, and ensure participation, transparency, and accountability in vaccine research, development, and manufacturing.
To All Governments
-
Fund efforts to develop and manufacture Covid-19 vaccines for the world, particularly for low- and middle-income countries, including through participating in the COVAX Facility and other efforts to pool procurement and equitably allocate vaccines worldwide.
-
Commit to sharing knowledge, intellectual property (IP), and data related to Covid-19 vaccines, therapeutics, and diagnostics, by endorsing the World Health Organization’s (WHO) COVID-19 Technology Access Pool (C-TAP) and implementing the “Solidarity Call to Action to realize equitable global access to COVID-19 health technologies through pooling of knowledge, intellectual property and data.”
-
Collectively map out the global capacity to manufacture the needed supply of Covid-19 vaccines, and cooperate to develop a strategy to fund and support the creation of additional manufacturing capacity especially in low- and middle-income countries to ensure rapid and diversified large-scale production.
-
Pledge not to sign bilateral deals with vaccine developers to prebook future vaccine doses in ways that undermine equitable global allocation according to public health need.
-
Support India and South Africa’s October 2020 proposal at the World Trade Organization to waive some provisions of the Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS Agreement) and use TRIPS flexibilities at the country-level.
-
Urgently introduce legislation to provide expedited compulsory licenses for all Covid-19 health products, including vaccines.
-
Urgently introduce legislation or other government regulations to cap the prices of all Covid-19 health products, including vaccines.
To Governments on the Boards of Gavi and CEPI, and those Participating in the COVAX Facility
-
Call on Gavi and CEPI to endorse and design the COVAX Facility in a way that is aligned with the WHO’s C-TAP.
-
Call on Gavi and CEPI to publish all agreements with vaccine developers and manufacturers.
-
Call on Gavi and CEPI to attach conditions to their funding related to technology transfers; open and non-exclusive licensing; and transparent and affordable pricing, verifiable by independent third-party audits that are made public.
-
Call on Gavi and CEPI to collectively map and publicly disclose all vaccine candidates in the COVAX Facility’s portfolio and their respective global supply chains. This should include the names of developers, funders, technologies used, manufacturers, manufacturing capacity booked and by whom.
-
Call on Gavi and CEPI to develop a process for and regularly consult with a diverse set of civil society representatives and groups, including those with varied expertise on access to medicines, IP barriers, and human rights. Publish reports from civil society consultations and meetings.
To Governments Funding Covid-19 Vaccines
-
Commit to transparency around Covid-19 vaccine funding by taking the following steps:
-
Create a public database of all contracts for funding related to Covid-19 vaccine research, development, manufacturing, or distribution;
-
Develop a process for and regularly consult with a diverse set of civil society groups and representatives with expertise on a range of public health and human rights issues. Publish reports from civil society consultations and meetings.
-
- Attach concrete conditions to all funding for Covid-19 vaccine research, development, manufacturing, or distribution requiring technology transfer, that is, recipients should share all IP, data, and other know-how to enable mass manufacturing of successful vaccine candidates. Specifically, ensure agreements to fund Covid-19 vaccines, at a minimum, require recipients to provide open, non-exclusive, and royalty-free licensing for pre-existing IP owned and any future owned IP modeled on the “Open COVID Pledge.”
- Attach concrete conditions to all vaccine funding to ensure vaccines are priced transparently and to prioritize affordability and minimize any debts for low- and middle-income countries, and not profits for vaccine-developers. Specifically, ensure agreements to fund Covid-19 vaccines, at a minimum, require that compliance with vaccine price commitments is verified through an independent third-party financial audit and the audit report be published.
- Commit to regular and rigorous independent, third-party auditing around terms and conditions of Covid-19 vaccine funding agreements. Publish detailed reporting on the findings and recommendations of these audits to enable external stakeholders to monitor outcomes.
To UN Organizations
-
Create opportunities and mechanisms for meaningful, sustained civil society participation in all global vaccine access initiatives. Publish reports from civil society consultations and meetings.
-
Call on governments, companies, research institutes, universities, multi-stakeholder initiatives and other entities involved in vaccine research and development to endorse the WHO’s C-TAP and join the “Solidarity call to action to realize equitable global access to COVID-19 health technologies through pooling of knowledge, intellectual property and data” and to commit to transparency by reporting regularly and publicly on their efforts and publishing agreements and other documents related to their work.
-
Take urgent measures to develop a roadmap to implement the WHO’s C-TAP in a time-bound manner and the participation of low- and middle-income countries and civil society groups with expertise on access to medicines, IP rights, and human rights.
-
Support India and South Africa’s October 2020 proposal to waive some provisions of the TRIPS Agreement and support individual country-level use of TRIPS flexibilities.
-
Take measures to promote and facilitate technology transfers of any successful Covid-19 vaccine to make it as widely available as possible.
-
Take measures to ensure any successful vaccine is priced affordably and made accessible to all vulnerable populations.
-
Provide technical support to governments to introduce legislation governing compulsory licenses for Covid-19 health technologies, including vaccines.
-
Develop a special searchable patent database for all Covid-19 patents relevant for vaccines, treatments, and testing.
Acknowledgments
This report was researched and written by Aruna Kashyap, senior counsel in the Business and Human Rights Division at Human Rights Watch, and Margaret Wurth, senior researcher in the Children’s Rights Division at Human Rights Watch. The report also drew on research conducted by Andrea Carvalho, senior research assistant with the Americas and Environment and Human Rights Divisions; Maham Javaid, fellow in the Middle East and North Africa Division; Carine Kaneza Nantulya, advocacy director in the Africa Division; Aya Majzoub, Lebanon and Bahrain researcher; Kathleen Page, M.D., associate professor of medicine at Johns Hopkins School of Medicine; Charbel Salloum, senior research assistant in the Middle East and North Africa Division; Jessica Tueller, intern in the Americas Division; and the Great Lakes researcher in the Africa Division. Brian Root, senior quantitative analyst, helped analyze the data on government funding for vaccines.
Sarah Saadoun, senior researcher in the Business and Human Rights Division, edited the report.
Joseph Amon, health and human rights consultant; Heather Barr, interim co-director of the Women’s Rights Division; Michael Garcia Bochenek, senior counsel to the Children’s Rights Division; and Akshaya Kumar, director of Crisis Advocacy and Special Projects reviewed and commented on the report. Aisling Reidy, senior legal adviser, provided legal review, and Babatunde Olugboji, deputy program director, provided program review.
Production assistance was provided by Namratha Somayajula, senior associate in the Business and Human Rights Division; Fitzroy Hepkins, senior administrative manager; and Jose Martinez, administrative officer.
Human Rights Watch gratefully acknowledges the time and insights offered by the health workers and many experts who guided our research and informed and shaped this report.